10/2025 – ADT Molecular has been awarded a tender by Hospital Sungai Buloh to supply our LyteStar™ MERS-CoV RT-PCR kit for the detection of Middle East Respiratory Syndrome (MERS) Coronavirus. The kit will be used to screen returning pilgrims from the Arabian peninsula with an indication of potential exposure.
AstronDX Technologies’ LyteStar™ MERS-CoV RT-PCR kit has achieved regulatory approval by the Malaysian Medical Device Authority (MDA) back in March 2023 as has since been utilized by a growing number of hospitals and public health laboratories. The awarded tender marks another milestone in our mission to deliver innovative quality products and impactful solutions that strengthen public health and biosecurity.
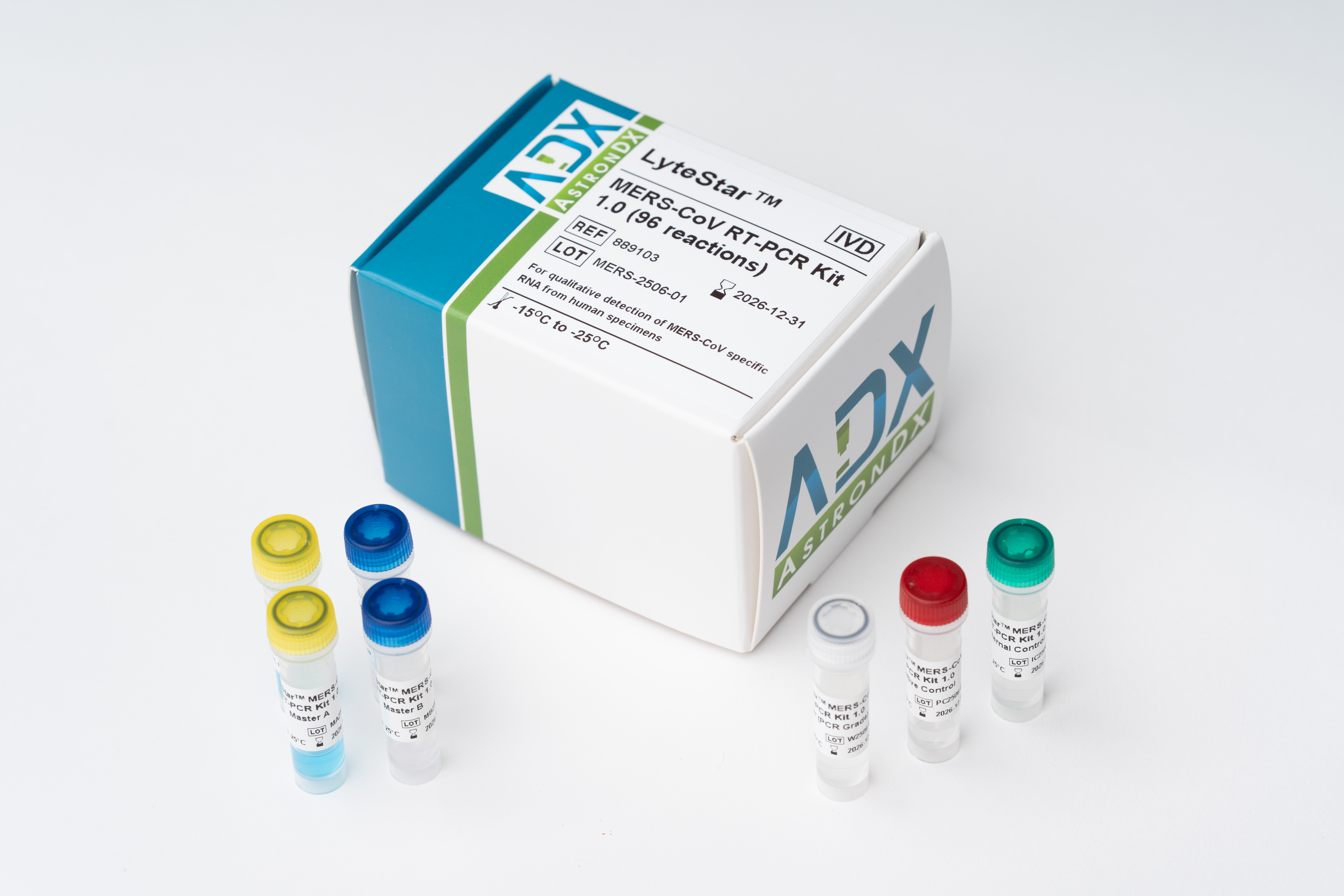
The LyteStar™ MERS-CoV RT-PCR Kit 1.0 is an in vitro diagnostic test system, based on real-time PCR technology, for the qualitative detection of Middle East Respiratory Syndrome (MERS-CoV) specific RNA. The LyteStar™ MERS-CoV RT-PCR Kit 1.0 consists of two assays in a single reaction tube, one targeting the upstream region of E gene (upE) and the other targeting the open reading frame 1a gene (Orf1a) of the MERS-CoV genome. The LyteStar™ MERS-CoV RT-PCR Kit 1.0 includes a heterologous amplification system (Internal Control) to identify possible RT-PCR inhibition and to confirm the integrity of the reagents in the kit.